
Fighting the superbugs
We're facing up to the challenge of antimicrobial resistance and ensuring that we protect modern medicine for future generations.
Without urgent action, as many as 10 million people each year could die as a result of AMR. To stop the spread of drug-resistant illness and disease, a new approach is desperately needed.
Antimicrobial resistance (AMR) is a global threat to human health, killing 1.2 million people in 2019 alone.
To stop the spread of drug-resistant illness and disease, urgent action is needed.
Antimicrobial resistance refers to the ability of microbes, such as bacteria, to withstand the drugs designed to kill them.
This ability is a biological change within the microbes themselves. Resistance develops through spontaneous genetic mutation in bacteria; once it occurs, those resistant bacteria survive antibiotic treatment and reproduce, passing their resistant genes on to successive generations. Over time, this creates 'superbugs' - populations of microbes that are extremely difficult, or even impossible, to treat with medicines.
When microbes become resistant to treatment, the most immediate consequence is that infections that were once straightforward to cure can become life-threatening.
Ordinary illnesses that once required a simple prescription can lead to prolonged hospital stays, or even fatalities. Common procedures that carry a risk of infection such as surgery, chemotherapy, or even childbirth become significantly more dangerous when the antibiotics used to prevent or treat resulting infections no longer work.
At a broader level, resistant infections place enormous strain on healthcare systems, requiring longer hospital stays, more expensive second- or third-line drugs, and intensive care that would otherwise be unnecessary.
If we do not rise to this challenge, it is entirely possible that we could see a return to a situation where 40 per cent of the population die prematurely from infections we cannot treat.
AMR is not a new phenomenon, but it is an urgent one.
The potential for microbes to become resistant over time was understood almost as soon as antibiotics were discovered. When Alexander Fleming accepted the Nobel Prize for the discovery of penicillin in 1945, he explicitly warned that misuse of the drug could lead to resistant bacteria.
What has changed is the scale and urgency of the problem. Decades of overuse and misuse of antibiotics in human medicine, agriculture, and livestock farming have dramatically accelerated the pace at which resistance develops and spreads. While resistance rises, the pipeline of new antibiotics has stalled. There have been no new antibiotics in more than 30 years, meaning we are depleting our existing arsenal faster than we are replacing it.
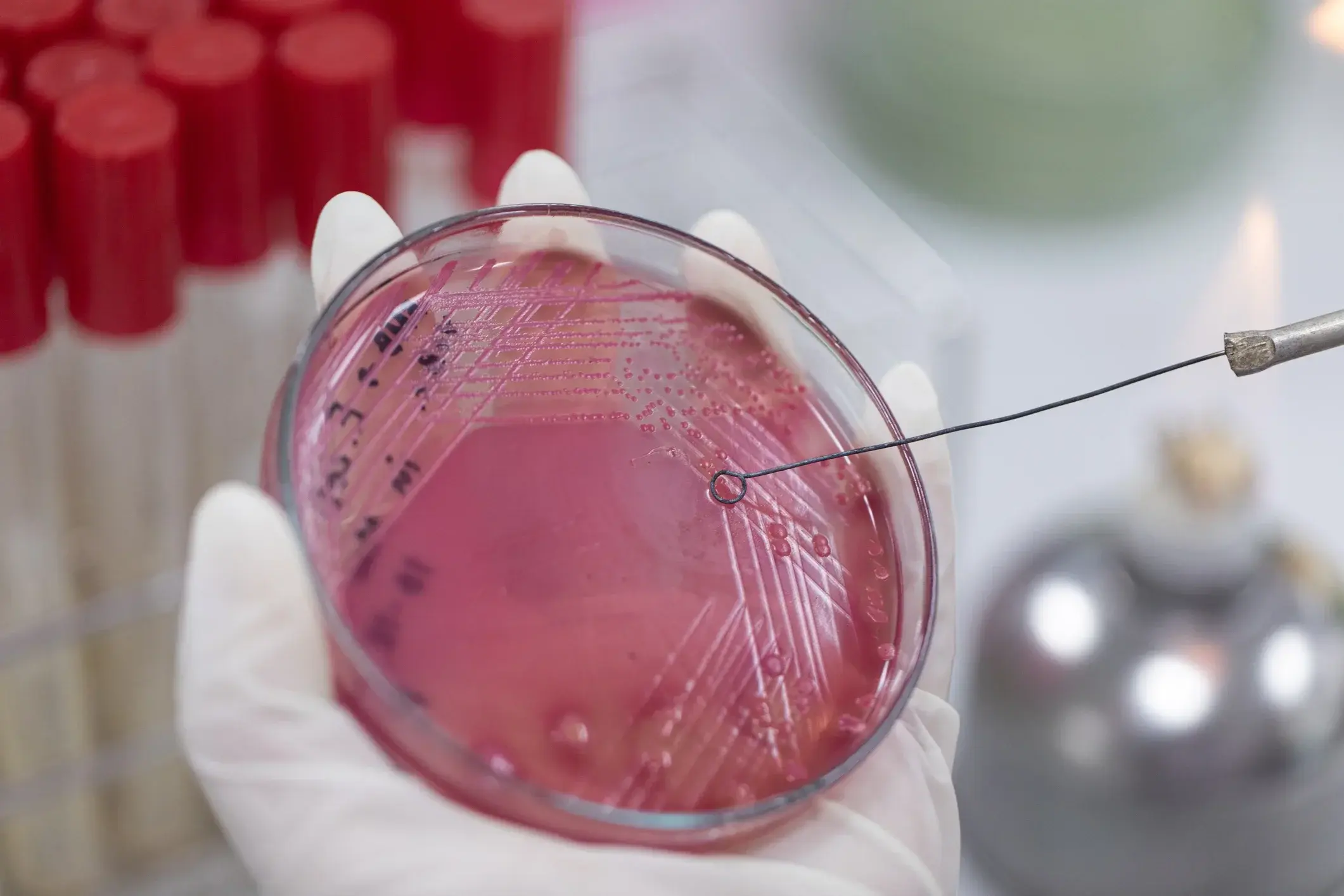
It is important to be clear that AMR cannot be stopped entirely as it is a process of natural selection. Resistant mutations will always occur; that is simply how evolution works.
What we can do, however, is try to outsmart the superbugs.
We can slow the process of resistance significantly and work on methods of reducing reliance on antibiotics. Research is a vital piece of this puzzle and investing in the development of new drugs and alternative therapies, ways to boost natural immunity, improving surveillance systems to track resistance patterns globally, alongside better infection prevention and sanitation, are all critical areas.
That’s where the Medical Research Foundation comes in.
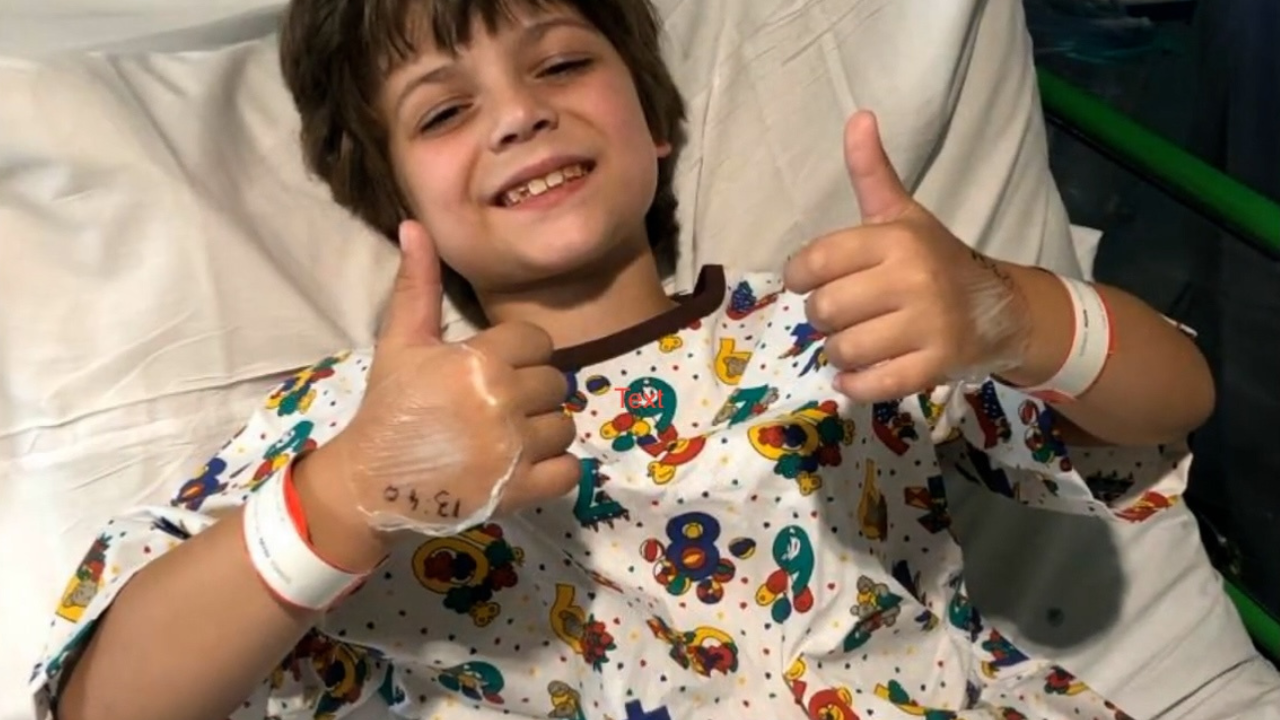
During his treatment for cystic fibrosis, eight-year-old Luke contracted an antibiotic-resistant infection in his lungs. Luke’s mother, Juliet, tells Luke’s story and the impact his condition had on their family.
Luke's storyin pioneering AMR research to fight the superbugs
The number of researchers working to tackle superbugs is small compared to the scale of the challenge. Many of the most important questions remain unanswered because they don’t fit neatly into traditional ways of funding and carrying out research.
Drug resistant superbugs affect the way we practise medicine, how we produce food, how we use medicines, and how we understand behaviour and systems. Yet research is often funded in silos, making it harder to bring the right ideas and the right people together.
We take a broader view; bringing together scientists, clinicians and researchers from very different fields, encouraging them to ask new questions and look at the problem from unexpected angles.

We committed £4 million to create the UK’s only national multi-disciplinary PhD training programme in antimicrobial resistance.
Find out more
In 2019, we funded outstanding scientists working on problems related to AMR through our Emerging Leaders Prize.
Find out more
We're inviting researchers in the field of antimicrobial resistance research to apply for funding. Apply before 2 July 2026.
Learn more and apply